Take samples – but do it right!

Viscosity of Liquids
 The right pump for each liquid
The right pump for each liquid
Pumps are used in widely different fields of application. Bürkle offers a
large number of different versions. However, when choosing a suitable
pump several criteria must be considered. The most important are the
viscosity, structure and density of the liquid. How the liquid behaves
under the influence of external influences, e.g. variations in
temperature and pressure, is also important.
Viscosity – what actually is viscosity?
The viscosity characterizes the flow behavior of a liquid.
High-viscosity liquids are thick, low-viscosity liquids thin. This
viscosity results from intermolecular forces; these are also known as
the internal friction. This internal friction decreases as the
temperature increases and increases as the temperature decreases. The
dynamic viscosity is given in Pascals, for low-viscosity liquids in
mPas. It is defined as the force in newtons that is necessary to
displace a liquid layer of 1 m2 and 1 m height along a boundary parallel
to a second layer at a speed of 1 m per second.
Viscosity using motor oil as an example
Everyone has heard of the viscosity terms for motor oil. This
classification is based on the flow behavior of the motor oil as a
function of the temperature. In Winter low-viscosity oils are required.
In Summer the oil becomes hotter, but it must still remain sufficiently
thick. For example: SAE 5W-40. The first number defines the viscosity at
0 °F (-18 °C), the second for a temperature of 210 °F (99 °C). An oil
with the classification 5W (W=Winter) is therefore thinner than a 15W
motor oil under cold conditions.
Typical viscosity values in mPas at 20 °C
| Kerosene |
0.65
|
|
Blood (37°C)
|
4 - 25
|
|
Olive Oil
|
~ 100
|
| Water |
1.0 |
|
Coffee Cream
|
~ 10
|
|
Hydraulic Oil
|
~ 120
|
| Mercury |
1.5 |
|
Glycol |
40 |
|
Yogurt |
~150 |
| Grape Juice |
2 - 5
|
|
Condensed Milk |
80 |
|
Motor Oil 15W 40
|
390 |
top of page
Take samples the right way
Correct sampling is very important in the context of quality assurance. It is essential if you want to have reliable and accurate data about the quality, properties or composition of a specific material.
You can carry out very precise material analysis using the most modern techniques. However, if a mistake is made in sampling then even these modern techniques are useless because the initial sampling error is carried through all the steps of the analysis and incorrect results will be obtained. This means that a correct statement about the properties of a substance can only be made if the samples are taken and handled with the same care and accuracy as is used in the subsequent analysis. It all comes down to the fact that the properties and composition of the sample itself must correspond exactly to those of the material from which it has been taken.
top of page
Take samples, but how?

A few basic steps must be observed when taking samples. It is recommended that you operate according to an internal company procedure, which should be a fixed component of the quality standards of every company.
All relevant sampling steps should be written down and carried out accordingly.
Sampling site
The site where sampling is carried out must be as clean as possible. If this is impossible due to external factors then, at the very least, care should be taken that clean equipment and auxiliary devices are used.
During the sampling process care must be taken that no external factors alter the sample. Even small impurities could falsify results.
Detergents that are used for cleaning the sampler and sample packaging can also alter results.
top of page
Take samples, but who?
Skilled workers
Only skilled workers with special training should take the samples. Sampling has to be carried out exactly in accordance with given guidelines. Skilled workers are able to recognize altered on-site conditions and react quickly to minimize the risk of sample contamination. A skilled worker recognizes malfunctions and changed conditions more quickly and analyzes them accordingly.
top of page
Take samples, but using what?
 Sampling equipment
Sampling equipment
The selection of the correct equipment is subject to many criteria. It depends on the material properties as well as the type of sampling, the containers in which the material is located, and the frequency of sampling. In addition to individual requirements, all equipment should fulfill the basic principles explained below.
Material
The equipment should be made of inert materials from which nothing can escape to contaminate the sample. Of course, this depends on the corrosiveness of the medium to be sampled. As a rule, equipment made of stainless steel and high quality synthetic or plastic material – especially fluoropolymers – is well suited.
Cleaning
It should be easy to clean the equipment quickly and – under certain conditions – to sterilize it. Depending on your needs, sterile-packed disposable samplers could be appropriate; these can be disposed of after use.
Processing
Equipment that is reused should not have anygrooves or crevices. Residual material collects cannot be easily removed; cross contamination can be a concern.
Surfaces
Equipment with especially smooth surfaces has proved to be especially well suited, for example, electropolished stainless steel or fluoropolymers. Such materials minimize deposits and greatly simplify cleaning. When selecting sampling equipment , you should take care to check that it is properly finished both inside and outside.
Sampling techniques - Physical and analytical sampling
A distinction is made between physical and analytical sampling. In physical sampling, the bulk density, material distribution, material properties and – when required, coloring or contamination are determined. It is especially important in this case that the sampler does not damage or destroy the material while withdrawing it. Also, keep in mind that a sample taken on site still has to be transported. You must also take care during transport and packing that the sample reaches the laboratory non-contaminated. In the analytical method, special attention must be paid to purity in addition to the aspects listed above.
On the other hand, the material properties can be disregarded sometimes, because the sample will be specially processed. In this case equipment can be used which changes the physical properties of the material.
top of page
Sampling techniques
 Multilayer sample
Multilayer sample
A sample is taken through all material layers to provide a representative cross-section.
Targeted sample
A targeted sample is taken from a pre-determined point. This is done to determine the material state precisely at this point or several targeted samples are taken over a larger area or in a large volume container. All target-point samples averaged together provide a representative cross-section of the material content.
Completion of the sampling procedure
After the sample has been withdrawn, the sample must be packed for transport. Clean sampling containers connected directly to the sampling equipment – without intermediaries – are ideal. The sample has no contact with the outside environment and is packed for transport at the same time. The same conditions relating to the sampling equipment apply to packaging, storage, and transport. Here too you should pay attention to the cleanliness of the packaging material and the risk of contamination from escaping substances. The containers are then labeled and taken to a laboratory – if necessary in coolers. Coolers with temperature monitors are ideal, to ensure that peak storage temperature is maintained.
Tip
If you take samples from sacks or bags, the insertion points can be reclosed with special labels. You can write on these labels, which will even stick firmly onto soiled surfaces.
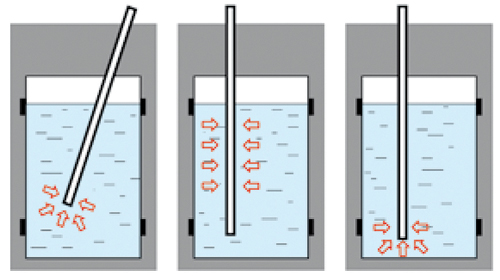
|
Point Sampling
|
Cross-Sectional Sampling
|
Bottom Sampling |
top of page
Contact us via e-mail (
[email protected]) or telephone 800-334-7585 with any further questions.